 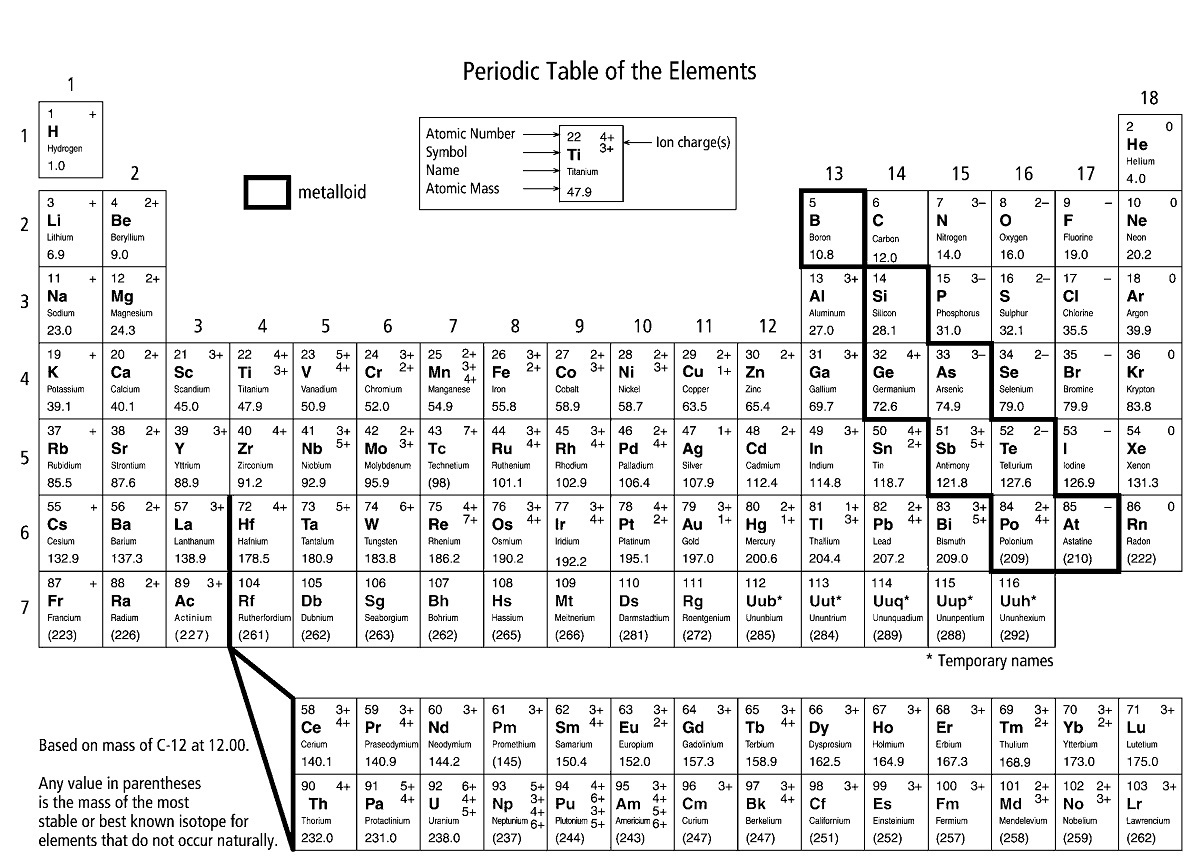
For example, carbon is in the same group as silicon as they both exhibit similar chemical properties. The result is the elements that are arranged in the same groups share similar chemical properties. These periodic changes correspond to periodic changes in the chemical behavior of elements as you go along the row. “What the ocean was to a child, the periodic table is to the chemist.” - Karl Barry Sharpless For example, elements on the leftmost part of the rows have few exterior electrons in their outer shells, and the number increases as you go from left to right. Elements in arranged in a period exhibit recurring chemical properties as you go from left to right. The periodic table is organized in a matrix of horizontal rows and vertical columns called periods and groups, respectively. Each cell in the periodic table has a symbol for the element, the atomic number of the element which corresponds to the number of protons in an atom of that element, and the average atomic weight of that element. What Is The Periodic Table?įirst some general facts about the periodic table. One of the most common ways of grouping elements in the periodic table are groupings based on general element-type, such as by the categories, metal, nonmetal, and metalloid. Because there are a number of ways to classify the elements, there exist a number of color-coding schemes we could use to point out specific properties that groups of elements share in common. Elements in the periodic table can be color-coded together according to atomic number, electron configuration, and recurring chemical properties. The color-coded periodic table is a tabular representation of the different chemical elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
